SURF 2022: Feedback control system in synthetic cells: Difference between revisions
No edit summary |
No edit summary |
||
| Line 3: | Line 3: | ||
Synthetic cells are non-living entities that may offer more advantages than living cells while implementing synthetic biological circuits due to lack of mutation and replication. Compartmentalizing different subsystems such as ‘sensor’, ‘actuator’ may provide the modularity required to build a complex bioengineered systems. Further, communication between synthetic cells and synthetic and living cells is vital to realize various bioengineering problems [1, 2]. This communication is often done with quorum signaling molecules [3]. A reasonable approach to move forward is by developing feedback or feedforward networks involving synthetic cells, which might solve more advanced synthetic biology problems. | Synthetic cells are non-living entities that may offer more advantages than living cells while implementing synthetic biological circuits due to lack of mutation and replication. Compartmentalizing different subsystems such as ‘sensor’, ‘actuator’ may provide the modularity required to build a complex bioengineered systems. Further, communication between synthetic cells and synthetic and living cells is vital to realize various bioengineering problems [1, 2]. This communication is often done with quorum signaling molecules [3]. A reasonable approach to move forward is by developing feedback or feedforward networks involving synthetic cells, which might solve more advanced synthetic biology problems. | ||
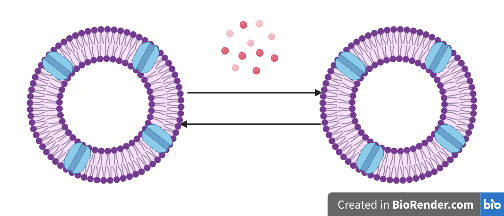
In traditional engineering systems, negative feedback control plays an important role in maintaining a desired level of output and achieving robustness against perturbations. In case of biomolecular systems, the | In traditional engineering systems, negative feedback control plays an important role in maintaining a desired level of output and achieving robustness against perturbations. In case of biomolecular systems, the negative feedback mechanism can be demonstrated by negative autoregulation of a species through phosphorylation-dephosphorylation type reactions. To realize robust perfect adaptation, integral feedback control motif is needed additionally. A negative feedback control strategy has been implemented to regulate rates of RNA production in case of genelet circuits [4] and in a robust growth rate control application in living cells through integral feedback control [5]. A similar feedback control strategy should be possible to implement in case of synthetic cells as well (Fig. 1). | ||
The broad goal of this summer project will involve modeling and implementing a feedback control system in synthetic cells. In particular, the robustness of a negative integral feedback control system against perturbations and whether the modularity provided by synthetic cells gives an advantage can be investigated. | The broad goal of this summer project will involve modeling and implementing a feedback control system in synthetic cells. In particular, the robustness of a negative integral feedback control system against perturbations and whether the modularity provided by synthetic cells gives an advantage can be investigated. | ||
Latest revision as of 23:18, 29 December 2021
Synthetic cells are non-living entities that may offer more advantages than living cells while implementing synthetic biological circuits due to lack of mutation and replication. Compartmentalizing different subsystems such as ‘sensor’, ‘actuator’ may provide the modularity required to build a complex bioengineered systems. Further, communication between synthetic cells and synthetic and living cells is vital to realize various bioengineering problems [1, 2]. This communication is often done with quorum signaling molecules [3]. A reasonable approach to move forward is by developing feedback or feedforward networks involving synthetic cells, which might solve more advanced synthetic biology problems.
In traditional engineering systems, negative feedback control plays an important role in maintaining a desired level of output and achieving robustness against perturbations. In case of biomolecular systems, the negative feedback mechanism can be demonstrated by negative autoregulation of a species through phosphorylation-dephosphorylation type reactions. To realize robust perfect adaptation, integral feedback control motif is needed additionally. A negative feedback control strategy has been implemented to regulate rates of RNA production in case of genelet circuits [4] and in a robust growth rate control application in living cells through integral feedback control [5]. A similar feedback control strategy should be possible to implement in case of synthetic cells as well (Fig. 1).
The broad goal of this summer project will involve modeling and implementing a feedback control system in synthetic cells. In particular, the robustness of a negative integral feedback control system against perturbations and whether the modularity provided by synthetic cells gives an advantage can be investigated.
Familiarity with ordinary differential equations, coding (python), and standard wetlab procedures will be helpful.
References:
[1] Adamala, K. P., Martin-Alarcon, D. A., Guthrie-Honea, K. R., & Boyden, E. S. (2017). Engineering genetic circuit interactions within and between synthetic minimal cells. Nature chemistry, 9(5), 431-439. https://www.nature.com/articles/nchem.2644
[2] Mukwaya, V., Mann, S., & Dou, H. (2021). Chemical communication at the synthetic cell/living cell interface. Communications Chemistry, 4(1), 1-12. https://www.nature.com/articles/s42004-021-00597-w
[3] Lentini, R., Martín, N. Y., Forlin, M., Belmonte, L., Fontana, J., Cornella, M., ... & Mansy, S. S. (2017). Two-way chemical communication between artificial and natural cells. ACS central science, 3(2), 117-123. https://pubs.acs.org/doi/abs/10.1021/acscentsci.6b00330
[4] Franco, E., Giordano, G., Forsberg, P. O., & Murray, R. M. (2014). Negative autoregulation matches production and demand in synthetic transcriptional networks. ACS synthetic biology, 3(8), 589-599. https://pubs.acs.org/doi/abs/10.1021/sb400157z
[5] Aoki, S. K., Lillacci, G., Gupta, A., Baumschlager, A., Schweingruber, D., & Khammash, M. (2019). A universal biomolecular integral feedback controller for robust perfect adaptation. Nature, 570(7762), 533-537. https://www.nature.com/articles/s41586-019-1321-1