SURF 2015: Design space exploration of the violacein pathway in TX-TL
2015 SURF project description
- Mentor: Richard Murray
- Co-mentor: Shaobin Guo and Yong Wu
Project Description
This project aims to explore the possibilities of utilizing cell-free systems to resolve some of the challenges and difficulties in metabolic engineering. Challenges in studying metabolic pathway include feedback loop, bifunctional enzymes, and parallel reactions (Woolston, 2012). Furthermore, the precise control of enzyme expressions and activities in complex pathway is difficult to achieve. On the other hand, cell-free systems simulate controlled cell environment. Several key benefits of using cell-free systems as biomolecular breadboard include small reaction volume, protein expressions from linear pieces of DNAs, and simultaneous protein expressions of multiple pieces of DNAs (Sun, 2013). These unique properties open up the possibilities of modulating concentrations of DNA encoding individual pathway enzymes, testing the related effect on metabolite production, and exploring pathway design space.
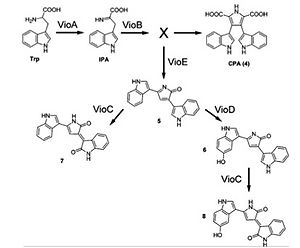
Violacein is a water-insoluble violet pigment that naturally exists in a few bacteria (Jiang, 2010 & Pantanella, 2007). Violacein has the potential applications in antibacterial, anti-trypanocidal, anti-ulcerogenic, and anticancer drugs (Balibar, 2006). The various enzymes involved in the pathway can lead to the production of byproducts that might interfere with quantification of violacein. The goal of the project is to characterize the violacein pathway using cell-free transcription and translational (TX-TL) system. Students will develop reliable quantification methods for violacein and take advantages of the TX-TL system to explore ways to improve violacein production.
Skills: Students interested in this project is required to have some basic microbiology laboratory experience ( Bi 1X, Bi 10, ChE/BE 130, or equivalent). Experience with either MATLAB, Python or a similar scientific programming package is strongly recommended. Other desired qualities include background in organic chemistry (Ch 41abc or equivalent) and experience with analytical techniques or separation methods (Ch 4a, Ch/ChE 9, Ch 24, ChE 62, or equivalent).
References:
Balibar, C. J. and C. T. Walsh (2006). "In Vitro Biosynthesis of Violacein from l-Tryptophan by the Enzymes VioA−E from Chromobacterium violaceum." Biochemistry 45(51): 15444-15457. [1]
Hoshino, T. (2011). "Violacein and related tryptophan metabolites produced by Chromobacterium violaceum: biosynthetic mechanism and pathway for construction of violacein core." Applied Microbiology and Biotechnology 91(6):1463-1475.
Jiang, P.-x., et al. (2010). "Reconstruction of the violacein biosynthetic pathway from Duganella sp. B2 in different heterologous hosts." Applied Microbiology and Biotechnology 86(4): 1077-1088.
Pantanella, F., et al. (2007). "Violacein and biofilm production in Janthinobacterium lividum." Journal of Applied Microbiology 102(4): 992-999.
Sun, Z. Z., et al. (2013). "Protocols for Implementing an Escherichia coli Based TX-TL Cell-Free Expression System for Synthetic Biology." (79): e50762.
Sun, Z. Z., et al. (2013). "Linear DNA for Rapid Prototyping of Synthetic Biological Circuits in an Escherichia coli Based TX-TL Cell-Free System." ACS Synthetic Biology.
Woolston, B. M., et al. (2013). "Metabolic Engineering: Past and Future." Annual Review of Chemical and Biomolecular Engineering 4(1): 259-288.